Live cell imaging
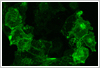
When imaging living cells on the confocal microscope it is important not just to keep the cells alive, but to make sure that they are close to their normal physiological state. Imaging dying cells or cells under stress will lead to misleading results. Imaging living cells requires careful consideration of a number of conditions:
Cellular structure and condition
Stress to cells must be minimised when transferring from the incubator to the microscope. This can be accomplished by good experimental procedures, design of the heated microscope stages and allowing cells time to recover before imaging.
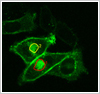 Temperature control
Temperature control
The use of heated stages and an objective heater are necessary to maintain a temperature of 370C in the cell chamber. Temperatures used will vary depending on the processes being studied.
Maintaining correct pH level
The correct pH level is vital for both the long-term viability of cultured cells and maintaining their correct physiological state. This can be achieved by imaging cells in HEPES buffered saline or in media with a CO2 incubator stage.
Photobleaching and reactive oxygen species (ROS)
Photobleaching is an ever-present problem when imaging living cells. Photobleaching results in the release of toxic ROS within the cells that react irreversibly with the excited dye producing non-fluorescent products.
Live cell imaging in the ICS
 We have considerable experience in imaging living cells within the ICS ranging from cultured cells to primary human cells. The confocal system in the ICS has numerous heated stages and an objective heater. The stages can accommodate for example, 35mm Mattek dishes, 22mm and 32mm round coverslips, Labtek coverslip chamber slides and a POC closed culture system. Many of the heated stages and inserts were manufactured to a high standard in the School Engineering Workshops. Information about the unit can be found at Medical Engineering Unit. All the coverslips need to be No1.5 thickness to allow the use of the high NA objective lenses. We have two water objectives available for live cell imaging a 40x/1.2W and a 63x/1.2W. For short-term studies we image cells in HEPES buffered saline. For longer term and overnight imaging we use the appropriate culture media (without phenol red) for the cells of interest and the CO2 incubator stage.
We have considerable experience in imaging living cells within the ICS ranging from cultured cells to primary human cells. The confocal system in the ICS has numerous heated stages and an objective heater. The stages can accommodate for example, 35mm Mattek dishes, 22mm and 32mm round coverslips, Labtek coverslip chamber slides and a POC closed culture system. Many of the heated stages and inserts were manufactured to a high standard in the School Engineering Workshops. Information about the unit can be found at Medical Engineering Unit. All the coverslips need to be No1.5 thickness to allow the use of the high NA objective lenses. We have two water objectives available for live cell imaging a 40x/1.2W and a 63x/1.2W. For short-term studies we image cells in HEPES buffered saline. For longer term and overnight imaging we use the appropriate culture media (without phenol red) for the cells of interest and the CO2 incubator stage.
Contact: Tim Self : 0115 823 0090